Speaker
Description
Removal of corrosion impurities from the molten Pb-16Li, which is a candidate breeder and coolant for the liquid metal breeding blanket system, is indispensable for the safe and efficient operation of the future fusion reactors. The corrosive nature of Pb-16Li in high temperature environment is responsible for the generation of corrosion impurities from the structural material. The deposition of corrosion impurities in the non-isothermal Pb-16Li loop may pose serious challenges (e.g. flow restriction due to plugging of flow path which further lead to the reduced heat exchange, generation of long lived activated corrosion products etc.) to the performance and safety of Pb-16Li breeding blanket system. In order to remove those corrosion impurities and activated corrosion products from the molten Pb-16Li, a purification component “Cold Trap” is generally recommended in the Pb-16Li loop system.
As a part of the development and testing of a Pb-16Li purification system, a wire mesh packing based prototype Cold Trap (CT) was designed, fabricated and installed in a purification loop with an objective to remove the impurities from ~ 260kg of Pb-16Li. In present work, we have assessed the functionality of CT in molten Pb-16Li system, continuously for a long duration of ~3500 hrs (~ 5 months). This long duration operation of CT in Pb-16Li system was imperative to test its reliability, since the recurrent replacement of the CT in the Pb-16Li system may not be feasible due to stringent operating conditions of fusion reactors. During the present experiment, we have achieved the reduction in concentration of impurities in Pb-16Li to their saturation solubility by passing the impure Pb-16Li through CT. The reduction in impurity level was also confirmed by the deposition of those dissolved impurities inside CT mesh packing in the needle shaped crystal formation. Some oxide depositions were also found inside the CT. Different characterization techniques such as SEM-EDX, XRD and ICP-AES were used for the characterization of the samples taken from the CT. The characterization results of deposited needle shaped crystal suggest the formation of Fe-Ni intermetallic while the XRD analysis of oxide deposition samples suggests the formation of Pb2O, Li2O and PbO.
Experimental details and main results: The prototype CT was installed in a bypass line of purification loop and a bleed (~10% nominal flow rate of the system) of Pb-16Li was passed through it for removal of dissolved corrosion impurities. In order to avoid freezing of Pb-16Li inside CT, Pb-16Li temperature inside the CT was maintained at ~260-270 oC which was ~25-35oC higher than the melting point of Pb-16Li ~235oC. The molten Pb-16Li inside the CT was cooled down to desired temperature using a temperature feedback controlled air cooling. During the experiment, samples of Pb-16Li were collected in-situ at different intervals to monitor the decrease in impurity concentration in Pb-16Li without disturbing the loop operation. In order to check the performance of CT for high solubility impurity like Ni, an impurity saturator was installed in bypass line of the purification loop to increase the concentration of Ni in Pb-16Li during the operation of system.
After completion of experiment, the corrosion impurities were found to be deposited in the needle shape crystalline form (~6-9 mm long needles and needle clusters) on the surface of the wire mesh packing of CT (refer Fig 1a). Small depositions of needle shaped crystal were also observed on the surface of CT outlet pipe (refer Fig 1b). The oxide impurities were found to be deposited on the internal walls of CT (refer Fig 1c).

The samples taken from the loop were analysed using ICP-AES techniques to determine their concentration in Pb-16Li. In the Pb-16Li samples taken after completion of experiment, the concentrations of Iron (Fe) and Nickel (Ni) which are the major corrosion impurities are found to be ~25ppm and ~925ppm, respectively. The concentration of these impurities are found to be near their saturation solubilities (~21 ppm for Fe and ~977ppm for Ni [refer 1, 2]) in the operating temperature range of CT (~260-270oC) and hence suggesting the deposition of excess impurities inside CT. In order to confirm this, the ICP-AES analysis of deposited crystals were performed. The concentration of Iron (Fe) and Nickel (Ni) in crystals deposited on wire mesh of CT are found to be ~74000 ppm and ~22000 ppm, respectively, which confirms the deposition of dissolved Fe and Ni impurities. The XRD and SEM-EDX analysis (refer Fig 2a and 2b) of crystals, deposited on the wire mesh packing of CT, also suggests the deposition of impurities in form of mainly Fe-Ni intermetallic.
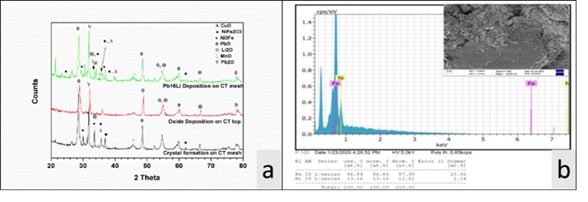
In addition to this, the ICP-AES analysis results of Pb-16Li samples also shows the decrease in the concentration of Copper (Cu) from ~190 ppm to ~50 ppm during the experiment. The oxide samples were analysed using XRD technique and the analysis results (refer Fig 2a) suggests the presence of PbO, Pb2O and Li2O.
References
[Ref 1] Investigation of models to predict the corrosion of steels in flowing liquid lead alloys, F. Balbaud-Celerier, F. Barbier, Journal of Nuclear Materials 289 (2001) 227-242
[Ref 2] The solubilities of nickel, manganese and chromium in Pb-17Li, Marten G. Barker and Tony Sample, Fusion Engineering and Design 14 (1991) 219-226
| Affiliation | Institute for Plasma Research, Bhat, Gandhinagar, India-382428 |
|---|---|
| Country or International Organization | India |
