Speaker
Description
Introduction: Since late 1970s, conventional Cr-Mo based ferritic/martensitic steels have been considered as candidate structural materials for fast and fusion reactors because of their higher swelling resistance and better thermal properties than austenitic steels 1. However, considering the damaging capacity of the high energy fusion neutrons, the Cr-W based reduced activation ferritic/martensitic (RAFM) steels are presents themselves as promising structural materials for fusion reactor applications 2. In RAFMS, alloying elements like Mo and Nb which produce long lived radioisotopes have been replaced with safer counterparts like W and Ta in order to ensure lower radioactive waste disposal time limits (~100 years) [1, 2]. In this regard, India has also developed its own RAFM steel (IN RAFMS) [9Cr–1.4W–0.06Ta–0.1C] altering the composition of the modified 9Cr-1Mo steel (P91) 3. IN RAFMS is the proposed structural material for the Indian Test Blanket Module (TBM) for ITER where liquid Pb-Li would be used as a coolant and tritium breeder 3. In general, substitution of W has been found to improve mechanical properties like impact and ductility of Cr-W steel (e.g. RAFMS) over the conventional Cr-Mo steels [4]. It is possible that the higher solid solution strengthening imparted by W is responsible for this behavior. Such observations highlighted the necessity to explore the effect of W addition over the compatibility of RAFMS with liquid metal/alloy coolants like Pb-Li which is one of its major qualifying criteria of structural materials for TBM applications. In this regard, a detailed investigation has been taken up at BARC, where Indian RAFM and P91 steels have been exposed to liquid Pb-Li under static and dynamic conditions at temperatures ranging 773 K to 823 K. The pre and post exposure surface properties and microstructures of both the above materials have been investigated thoroughly in order establish the role of W and Mo in the affecting the compatibility of ferritic/martensitic steels.
Results and Discussions: In the entire range of experimental conditions, IN RAFMS has been found to possess superior corrosion resistance than P91 in liquid Pb-Li. This has been observed both from weight loss measurements (Table 1) as well as microstructural investigations. To understand this difference in corrosion behavior, detailed investigations through X-Ray Photo Electron Spectroscopy (XPS) have been carried out on the pre exposed surface of the above materials. The results have shown that both P91 and IN RAFMS steel develops an ~ 8 nm thick air formed oxide layer over their surface containing a mixed oxide of iron and chromium [FeO/ (Fe, Cr)2 O3]. It was also observed that the oxide scale in IN RAFMS additionally contained tungsten oxides [WO2 and WO3] in the surface layer although molybdenum oxides were not observed in P91 under similar conditions. This is shown in Figure 2 which represents the XPS depth profile of W and Mo in IN RAFMS and P91 respectively.
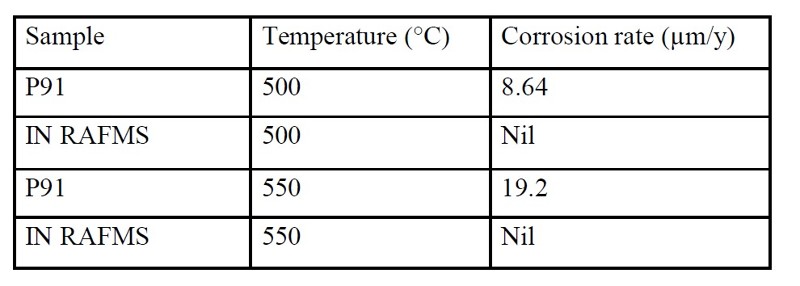
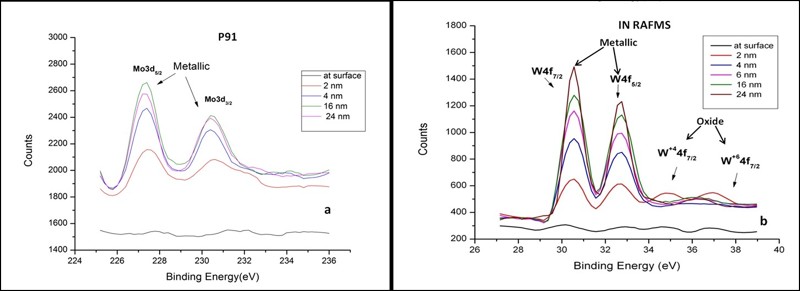
Analysis of Pb-Li exposed samples have revealed that Pb-Li attack over ferritic/martensitic steels initiates with the dissolution of the aforesaid surface oxide layer in Pb-Li (mainly in Li from thermodynamic considerations). This is accompanied by penetration of Pb-Li along the grain boundaries where the oxide layer is weaker due to the precipitation of chromium carbides (Cr23C6). The reactions for reduction of the Fe, Cr, W and Mo oxides (in case if present on P91) by Li has been simulated through Factsage software and the associated change in free energy (ΔG) has been evaluated at the experimental temperature 773 K. Such analysis has shown that the stability of tungsten oxides was highest among all the other oxides i.e. oxides of Fe, Cr as well as Mo both in Pb-Li environment as well as in air. This indicated that the presence of tungsten oxide in the surface layer of IN RAFMS is a major factor responsible for preventing its greater corrosion resistance to Pb-Li as compared to P91. On the other hand, analysis through Secondary Electron Microscopy (SEM) and Electron Back Scattered Diffraction (EBSD) have shown a competitively higher prior austenite grain size of IN RAFMS (~ 14 µm) in comparison to P91(~ 10 µm). The EBSD color maps of as-received microstructure of P91and IN RAFMS and are shown in Figure 3. The greater grain size reduces grain boundary attack during oxide dissolution phase and thus helps in imparting additional corrosion resistance to IN RAFMS.
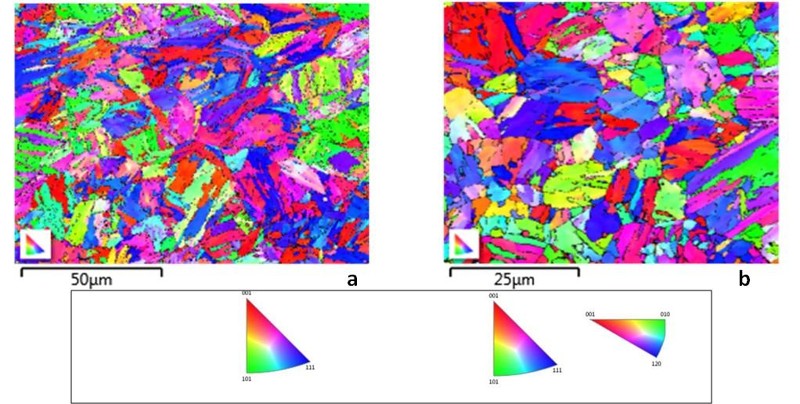
Conclusion: The present study has established that RAFM steels like IN RAFMS are more resistant to Pb-Li corrosion than the corresponding surrogate Cr-Mo steels like P91. Better corrosion resistance of IN RAFMS is attributed mainly to the presence of tungsten oxides in the passive layer which offers better stability and higher grain size in the case of IN RAFMS.Such an understanding of the effect of tungsten addition towards the corrosion process can help in engineering newer alloy compositions with improved Pb-Li corrosion resistance.
References:
- R. L. Klueh, K. Ehrlich and F. Abe, Journal of Nuclear Materials 191-194 (1992)116-124
- L. Tan, L. L. Snead, Y. Katoh, Journal of Nuclear Materials 478 (2016) 42-49.
- B. Raj, K. B. S. Rao, A.K.Bhaduri , Fusion Engineering and Design 85 (2010) 1460–1468
- R. L. Klueh and W.R. Corwin, J. Mater. Eng. 11 (1989) 169-175
| Affiliation | Materials Science Division, Bhabha Atomic Reserach Center, Mumbai |
|---|---|
| Country or International Organization | India |
